Tetramethylethylenediamine

| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
N,N,N′,N′-Tetramethylethane-1,2-diamine[1] | |
| Identifiers | |
3D model (JSmol)
|
|
| Abbreviations | TMEDA, TEMED |
| 1732991 | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.003.405 |
| EC Number |
|
| 2707 | |
| MeSH | N,N,N',N'-tetramethylethylenediamine |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 2372 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H16N2 | |
| Molar mass | 116.208 g·mol−1 |
| Appearance | Colorless liquid |
| Odor | Fishy, ammoniacal |
| Density | 0.7765 g mL−1 (at 20 °C) |
| Melting point | −58.6 °C; −73.6 °F; 214.5 K |
| Boiling point | 121.1 °C; 249.9 °F; 394.2 K |
| Miscible | |
| Acidity (pKa) | 8.97 |
| Basicity (pKb) | 5.85 |
Refractive index (nD)
|
1.4179 (20 °C)[2] |
| Hazards | |
| GHS labelling: | |
  
| |
| Danger | |
| H225, H302, H314, H332 | |
| P210, P280, P305+P351+P338, P310 | |
| NFPA 704 (fire diamond) | |
| Flash point | 20 °C (68 °F; 293 K) |
| Explosive limits | 0.98–9.08% |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
|
| Related compounds | |
Related amines
|
Triethylenetetramine |
Related compounds
|
|
| Supplementary data page | |
| Tetramethylethylenediamine (data page) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Tetramethylethylenediamine (TMEDA or TEMED) is a chemical compound with the formula (CH3)2NCH2CH2N(CH3)2. This species is derived from ethylenediamine by replacement of the four amine hydrogens with four methyl groups. It is a colorless liquid, although old samples often appear yellow. Its odor is similar to that of rotting fish.[4]
As a reagent in synthesis
[edit]TMEDA is widely employed as a ligand for metal ions. It forms stable complexes with many metal halides, e.g. zinc chloride and copper(I) iodide, giving complexes that are soluble in organic solvents. In such complexes, TMEDA serves as a bidentate ligand.
TMEDA has an affinity for lithium ions.[4] When mixed with n-butyllithium, TMEDA's nitrogen atoms coordinate to the lithium, forming a cluster of higher reactivity than the tetramer or hexamer that n-butyllithium normally adopts. BuLi/TMEDA is able to metallate or even doubly metallate many substrates including benzene, furan, thiophene, N-alkylpyrroles, and ferrocene.[4] Many anionic organometallic complexes have been isolated as their [Li(tmeda)2]+ complexes.[5] In such complexes [Li(tmeda)2]+ behaves like a quaternary ammonium salt, such as [NEt4]+.
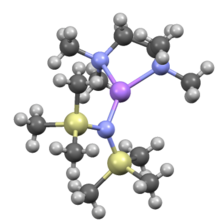
TMEDA adduct of lithium bis(trimethylsilyl)amide Notice that the diamine is a bidentate ligand.[6]
sec-Butyllithium/TMEDA is a useful combination in organic synthesis where the n-butyl analogue adds to substrate. TMEDA is still capable of forming a metal complex with Li in this case as mentioned above.
In molecular biology
[edit]TEMED is a common reagent in molecular biology laboratories, as a polymerizing agent for polyacrylamide gels in the protein analysis technique SDS-PAGE.[7]
Other uses
[edit]The complexes (TMEDA)Ni(CH3)2 and [(TMEDA)Ni(o-tolyl)Cl] illustrate the use of tmeda to stabilize homogeneous catalysts.[8] [9]

Related compounds
[edit]- H2NCMe2−CMe2NH2, also referred to as tetramethylethylenediamine.[10]
- Bis(dimethylamino)methane, Me2NCH2NMe2
References
[edit]- ^ "N,N,N′,N′-tetramethylethylenediamine – Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 16 September 2004. Retrieved 30 June 2012.
- ^ Lide, David R., ed. (2009). CRC Handbook of Chemistry and Physics (90th ed.). Boca Raton, Florida: CRC Press. ISBN 978-1-4200-9084-0.
- ^ "MSDS" (PDF).[permanent dead link]
- ^ a b c Haynes, R. K.; Vonwiller, S. C.; Luderer, M. R. (2006). "N,N,N′,N′-Tetramethylethylenediamine". In Paquette, L. (ed.). N,N,N′,N′-Tetramethylethylenediamine. Encyclopedia of Reagents for Organic Synthesis. New York: J. Wiley & Sons. doi:10.1002/047084289X.rt064.pub2. ISBN 0471936235.
- ^ Morse, P. M.; Girolami, G. S. (1989). "Are d0 ML6 Complexes Always Octahedral? The X-ray Structure of Trigonal Prismatic [Li(tmed)]2[ZrMe6]". Journal of the American Chemical Society. 111 (11): 4114–4116. doi:10.1021/ja00193a061.
- ^ Henderson, K. W.; Dorigo, A. E.; Liu, Q.-L.; Williard, P. G. (1997). "Effect of Polydentate Donor Molecules on Lithium Hexamethyldisilazide Aggregation: An X-ray Crystallographic and a Combination Semiempirical PM3/Single Point ab Initio Theoretical Study". J. Am. Chem. Soc. 119 (49): 11855. doi:10.1021/ja971920t.
- ^ Manns, Joanne M. (2011). "SDS-Polyacrylamide Gel Electrophoresis (SDS-PAGE) of Proteins". Current Protocols in Microbiology. 22. doi:10.1002/9780471729259.mca03ms22.
- ^ Shields, Jason D.; Gray, Erin E.; Doyle, Abigail G. (2015-05-01). "A Modular, Air-Stable Nickel Precatalyst". Organic Letters. 17 (9): 2166–2169. doi:10.1021/acs.orglett.5b00766. PMC 4719147. PMID 25886092.
- ^ Magano, Javier; Monfette, Sebastien (2015-04-17). "Development of an Air-Stable, Broadly Applicable Nickel Source for Nickel-Catalyzed Cross-Coupling". ACS Catalysis. 5 (5): 3120–3123. doi:10.1021/acscatal.5b00498.
- ^ Jackson, W.Gregory; Rahman, A.F.M.Mokhlesur; Wong, M.Anthony (2004). "Solvent Exchange, Solvent Interchange, Aquation and Isomerisation Reactions of cis- and trans-[Co(tmen)2(NCMe)2]3+ in Water, Me2SO and MeCN: Kinetics and Stereochemistry". Inorganica Chimica Acta. 357 (3): 665–676. doi:10.1016/j.ica.2003.05.010.


